According to the following figure, the magnitude of the enthalpy change of the reaction A + B → M + N in kJ mol^–1 - Sarthaks eConnect | Largest Online Education Community

Calculated Free Energies (G, kJ/mol) and Vibrational Frequencies (ν̃ ,... | Download Scientific Diagram
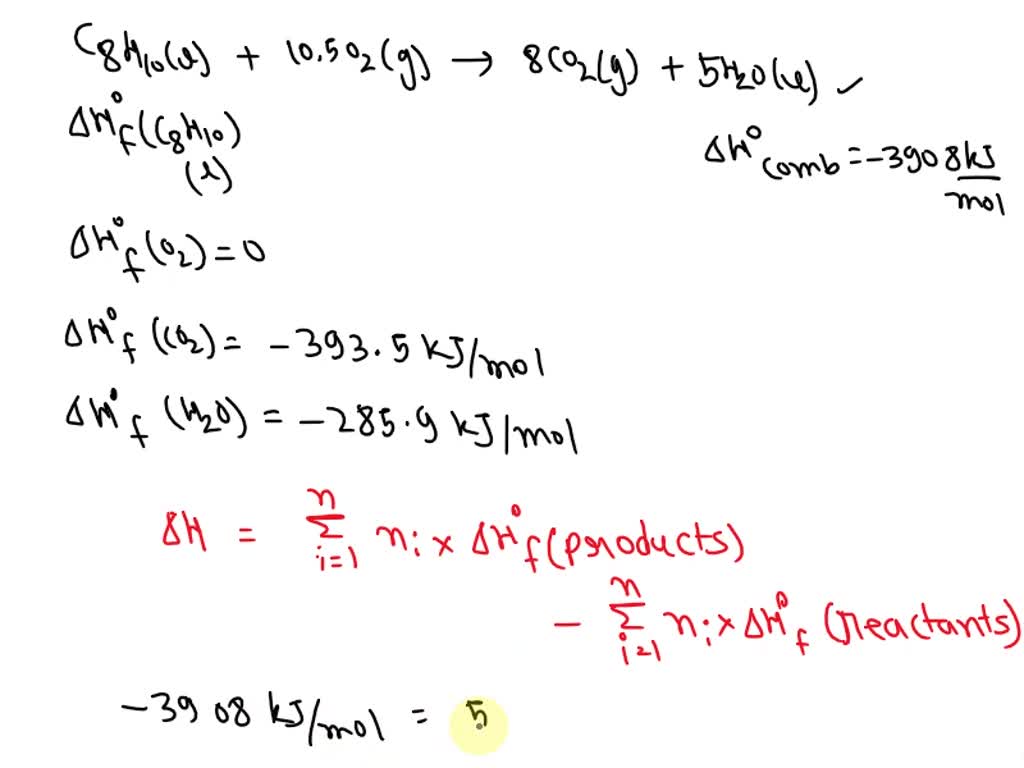
SOLVED: Physical chemistry Knowing that the standard enthalpy of combustion for xylene, CgH10(I), is -3908 kJ mol-1. Using HOf: H2O(l) = -285.9 kJ mol-1; CO2(g) = -393.5 kJ mol-1. CgH10(l) + 10.5O2(g)
The activation energy of a reaction is 75.2 kJ mol^-1 in the absence of a catalyst and it lowers to 50.14 kJmol^-1 with a catalyst. - Sarthaks eConnect | Largest Online Education Community

Enthalpy of formation of H,O, is: (1) +X, kJ mol-1 127-X, kJ mol (3) +X, kJ molt (4) -X, kJ mol Given that bond energies of H-Hand CHCl are 430 kJ moll

enthalpy - Why is it sometimes kJ only, and in other times kJ/mol? What's the difference? - Chemistry Stack Exchange

66. CH + 50, +300, + 4H,0 AH =-2220 kJ moF1 CH+H, CH, AH =-124 kJ mol-1 H+ +0, H,0 AH = -285 kJ mol-1 Then heat of combustion of CH (1)-2059 kJ mol-1 (2) 2059 kJ mo (3) -4118 kJ mol- (4) +4118 kJ mol-1






![Solved Given that Delta H degree r[H(g)] = 218.0 kJ. mol-1 | Chegg.com Solved Given that Delta H degree r[H(g)] = 218.0 kJ. mol-1 | Chegg.com](https://d2vlcm61l7u1fs.cloudfront.net/media/f73/f73ff219-6265-444d-89c1-620f5d2f2822/php7wff8s.png)